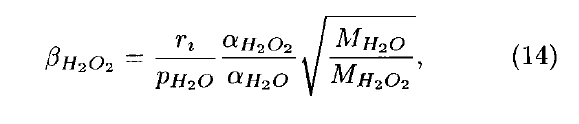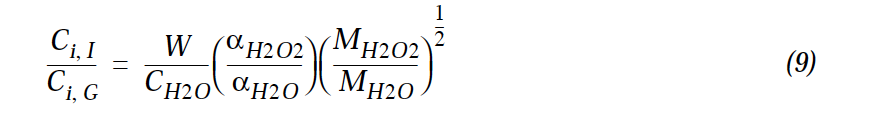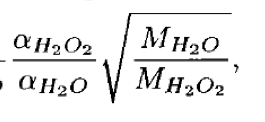Wet deposition
This page describes the current wet deposition scheme used in GEOS-Chem.
Overview
The Harvard Atmospheric Chemistry Modeling Group developed a wet deposition scheme (including scavenging of soluble tracer in convective updrafts, as well as rainout and washout of soluble tracers) for the GMI model. This scheme was then implemented into GEOS-Chem. Jacob et al [2000] describes the algorithm in full. This scheme is also described in Liu et al [2001].
A number of updates have been added since the implementation of the original wet scavenging algorithm. These include:
Allow both washout and rainout when precipitation forms
NOTE: This update was incorporated into GEOS-Chem v9-01-01.
Qiaoqiao Wang wrote:
- When there is new formation of precipitation in lower layer k, rainout will be applied to the whole precipitation area: max(Fk,Fk+1), considering the contribution of precipitation formation overhead. This will overestimate rainout effect when Fk+1 is much larger than Fk. Therefore, we now apply rainout effect to precipitation area Fk and washout effect to the area: max (0, Fk+1-Fk) in the same grid box.
Updates for aerosol scavenging efficiency
This update was tested in the 1-month benchmark simulation v9-01-03e and approved on 02 Feb 2012.
Qiaoqiao Wang wrote:
- The bulk below-cloud scavenging parameterization of Dana and Hales (k = 0.1P, where P is the precipitation rate mm h-1) used in the standard GEOS-Chem model integrates scavenging efficiencies over typical aerosol size distributions. This overestimates scavenging as it does not account for the preferential removal of the very fine and coarse particles over the course of the precipitation event, shifting the aerosol size distribution toward the more scavenging-resistant accumulation mode that accounts for most of aerosol mass. Now we use the below-cloud scavenging coefficients (k = a Pb constructed by Feng (2007, 2009)) integrated over accumulation mode for most aerosols and over coarse mode for coarse dust and sea salt.
--Bob Y. 15:50, 11 January 2011 (EST)
Updates for MERRA met fields
In GEOS-Chem v9-01-01, we have implemented an improved wet deposition scheme which uses the precipitation fields directly from the MERRA reanalysis product.
The wet deposition algorithm for GEOS-5 is relatively unchanged, except that we allow both rainout and washout to form within a grid box simultaneously. The wet deposition code has been partitioned into several new subroutines within wetdep_mod.F90 in order to allow for better compatibility with the wet deposition scheme used for the MERRA met fields.
--Bob Y. 14:12, 9 March 2011 (EST)
Add scavenging by snow
This update was tested in the 1-month benchmark simulation v9-01-03e and approved on 02 Feb 2012.
Qiaoqiao Wang wrote:
- For in-cloud scavenging by rain droplets, we assume 100% of water-soluble aerosols are rained out. But in the case of snow, only dust and hydrophobic BC are considered to be IN and then could be rained out. Note that HNO3 is also assumed to be rained out by snow as it forms a monolayer in ice crystal. The below-cloud scavenging coefficients are also higher for snow than for rain droplets.
--Bob Y. 14:14, 9 March 2011 (EST)
Impaction scavenging for hydrophobic BC and homogeneous IN removal
These updates were validated with 1-month benchmark simulation v11-01b and 1-year benchmark simulation v11-01b-Run0. This version was approved on 19 Aug 2015.
From Wang et al. [2014]:
- We modify the scheme by (1) scavenging hydrophobic aerosol (hydrophobic BC and OC) in convective updrafts, since this would take place by impaction [Ekman et al., 2004], and (2) scavenging water-soluble aerosol from cold clouds by homogeneous freezing of solution droplets at T < 237 K [Friedman et al., 2011]. We conducted 222Rn-210Pb simulation to test the general model representation of aerosol deposition and found a lifetime of tropospheric 210Pb aerosol against deposition of 8.6 days.
--Melissa Sulprizio 11:13, 26 November 2013 (EST)
Implementation of the species database
This update was validated with 1-month benchmark simulation v11-01d and 1-year benchmark simulation v11-01d-Run1. This version was approved on 12 Dec 2015.
We have created a new data structure—the [GEOS-Chem species database—that holds physical properties for each GEOS-Chem species]]. These physical properties include molecular weights, Henry's law constants, dry deposition parameters, and wet deposition parameters. The implementation of the species database will allow us to remove repetitive (and confusing) code, particularly in routines INIT_WETDEP, WETDEPID, COMPUTE_F, RAINOUT, and WASHOUT.
Implementation of the species database has begun in GEOS-Chem v11-01. This will be a multi-stage process. It should be noted that this will be a structural update, and will not change any science. But in the future, with the species database in place, updating physical properties for wet deposition will be much easier than it has been in prior GEOS-Chem versions. All that will be necessary is to update species physical properties in a single location, and they will be propagated to other locations in GEOS-Chem automatically.
As part of this update, the wet deposition module now calls the same Henry's law routines that are used by the HEMCO emissions component. This ensures that the same algorithm is used to compute the dimensionless Henry's law liquid/gas constant everywhere in GEOS-Chem.
--Bob Yantosca (talk) 21:45, 15 December 2015 (UTC)
Update SO2 scavenging in convective updrafts for consistency
Temporary fix to remove SO2 scavenging in convective updrafts
This update is slated for GEOS-Chem v11-02.
Duncan Fairlie wrote:
- It was indeed the instantaneous titration of H2O2 by SO2 and its subsequent scavenging as sulfate Which I deduced was leading to excessive removal of SO2 in wet convective updrafts, and consequent Deficits of both SO2 and sulfate in the UTLS above the Asian monsoon.(It’s a problem also in GOCART, Which I think is the origin of the G-C approach).
- My solution (which may or may not be valid) was to to treat SO2 as partially soluble, via the effective Henry’s Equilibrium, so that only the fraction of SO2 in the cloud condensate would be subject to wet scavenging. i.e. Treat SO2 in the same way as H2O2 are CH2O are currently treated in COMPUTE_F routine (see attached wetscav module). Note, I am not considering subsequent in-cloud sulfate production from H2O2 or O3 here. That is done in sulfate_mod. I’m essentially focusing on the fraction of SO2 taken up in the cloud water, and hence subject to wet scavenging.
- As I say, I am now treating this quite separately from the sulfate production from in-cloud SO2 oxidation by H2O2 And O3 that is done in the sulfate_mod module. In sulfate_mod, the solubility of SO2 in cloud water IS Treated using the Henry’s Law constant; the sulfate produced is then subject to wet scavenging as aerosol.
- So, it seems to me, in the standard code, that there is potential multiple counting of SO2 loss in wet Convective updrafts - instantaneous titration of H2O2 and subsequent scavenging, and also what’s going on in sulfate_mod where the dissolved SO2 is oxidized by H2O2 and O3 to sulfate which is then scavenged.
- What you propose sounds like a more appropriate solution. My approach seemed quick and simple, an improvement I think, but perhaps not the correct thing to do.
Final solution to account for SO2 oxidation in updrafts
Daniel Jacob wrote:
- I agree with Duncan that the current formulation in the standard code double-counts SO2 oxidation in precipitating clouds since this oxidation is done in both sulfate_mod and in scavenging. That should be a relatively small effect since 90% of clouds don't precipitate. However, I think it's essential to separately account for SO2 oxidation in convective updrafts, since otherwise SO2 scavenging in these updrafts will be negligible and all the SO2 will be detrained at the top. We should place a kinetic limitation on SO2 oxidation in convective updrafts, following the formulation in sulfate_mod, rather than assume instantaneous titration of SO2 by H2O2 as is applied at present. This would allow more SO2 to be detrained at the top of the updraft while still recognizing the potential for SO2 scavenging due to oxidation in the updraft.
- In the meantime, should we implement [Duncan's] current code that doesn't allow for SO2 scavenging in convective updrafts? That should be better than what we have right now (100% scavenging).
--Melissa Sulprizio (talk) 21:51, 10 November 2015 (UTC)
Validation
See Liu et al [2001].
Previous issues that have been resolved
Removal of obsolete variables NSOL and IDWETD
These updates were included in v11-02a and approved on 12 May 2017.
The following variables in the wet deposition module GeosCore/wetscav_mod.F were replaced with fields from the State_Chm object:
| Variable | Removed | Added |
|---|---|---|
| NSOL | DO N = 1, NSOL, etc. | DO N = 1, State_Chm%nWetDep |
| IDWETD | N = IDWETD(NSOL), etc. | N = State_Chm%Map_Advect(N_WD) |
This update passed geosfp_4x5_tropchem and geosfp_4x5_standard difference tests on 17 Mar 2017.
--Bob Yantosca (talk) 19:15, 17 March 2017 (UTC)
Fix bug in GEOS-FP/MERRA/MERRA-2 re-evaporation calculation
This update was validated with the 1-month benchmark simulation v11-01i (approved 20 Oct 2016).
Viral Shah wrote:
- Please see attached the revised code (wetscav_mod.F) and a patch file (w.r.t. commit 2f641a) with the recommended bug fix ('fix1') for the GEOS-FP/MERRA re-evaporation calculation. The original code did not take into account the rate of re-evaporation of precipitation (GEOS-FP/MERRA field: REEVAPLSAN) when calculating the fraction of particles (and HNO3) re-suspended in a gridbox. The attached fix corrects this error.
- Test simulations using this fix resulted in an annual lifetime for Pb210 of ~8.3 days for GEOS-FP and MERRA (simulation performed by Bo Zhang). Bo also found better correspondence with aircraft observations in the test simulations.
- The specific changes included in the fix are:
- a) Declare and initialize the REEVAP array to hold the re-evaporation rate, and
- b) In the WETDEP_MERRA routine, pass REEVAP instead of QQ to the DO_WASHOUT_ONLY routine to calculate the fraction of precipitation re-evaporating. This fraction (ALPHA) is now calculated as: (REEVAP * BXHEIGHT)/PDOWN.
- c) REEVAP occasionally has negative values but with small magnitudes. We set these to zero.
- I would like to acknowledge again the work of Bo and Hongyu in evaluating the fix.
--Melissa Sulprizio (talk) 12:32, 18 July 2016 (UTC)
Now use the same Henry's law functions that are used in HEMCO
This update was validated with the 1-month benchmark simulation v11-01e (approved 04 Jan 2016).
The quantity L2G (mixing ratio of species in the liquid phase / mixing ratio of species in the gas phase) is used throughout the GEOS-Chem wet deposition module (GeosCore/wetscav_mod.F) to compute the fractions of soluble species lost to updraft scavenging, large-scale rainout, or large-scale washout.
In GEOS-Chem v11-01d and prior versions
In GEOS-Chem v11-01d and prior versions, routine COMPUTE_L2G used this algorithm:
! R = universal gas constant [L*atm/moles/K] REAL(fp), PARAMETER :: R = 8.2057e-2_fp ! INV_T0 = 1/298 K REAL(fp), PARAMETER :: INV_T0 = 1e+0_fp / 298e+0_fp ! Get Kstar, the effective Henry's law constant for temperature TK Kstar = Kstar298 * EXP( -H298_R * ( ( 1e+0_fp / TK ) - INV_T0 ) ) ! Use Henry's Law to get the ratio: ! [ mixing ratio in liquid phase / mixing ratio in gas phase ] L2G = Kstar * H2OLIQ * R * TK
where:
- KStar298 is the Henry's law solubility constant [M atm-1]
- H_298 is the Henry's law volatility constant [K]
- TK is the temperature [K]
- Kstar is the dimensionless Henry's law liquid/gas constant at temperature TK
- H2OLIQ is the liquid water content [cm3 H2O cm-3 air]
In GEOS-Chem v11-01e and later versions
The HEMCO emissions component update in GEOS-Chem v10-01 and later versions introduced a new module (GeosUtil/henry_mod.F) for computing the dimensionless Henry's law liquid/gas constant. This module contains two functions:
- CALC_KH: Computes KH, the dimensionless liquid/gas Henry's law constant, for a given species and temperature.
- CALC_HEFF: Applies a pH correction to KH (if the species is pH-dependent).
To ensure that all Henry's law computations in GEOS-Chem are done with the same algorithm, we have updated the COMPUTE_L2G routine to use the CALC_KH and CALC_HEFF functions, as shown below:
! Cast temperature to REAL*8 TK_8 = TK ! For wetdep, we assume a pH of 4.5 for rainwater pH = 4.5_f8 ! Calculate the Henry's law constant CALL CALC_KH( K0, CR, TK_8, KH, RC ) !--------------------------------------------------------------- ! NOTE: CALC_KH uses this formula, compare this to the ! formula from the old COMPUTE_L2G above: REAL*8, PARAMETER :: TREF = 298.15d0 ! [K ] REAL*8, PARAMETER :: R = 8.3144621d0 ! [J K-1 mol-1] REAL*8, PARAMETER :: ATM = 101.325d0 ! [mPa (!) ] ! Calculate Henry coefficient for given temperature KH = thisK0 * exp( thisCR * (1/TK - 1/TREF) ) * R * TK / ATM !--------------------------------------------------------------- ! Calculate effective Henry's law constant, corrected for pH ! (for those species that have a defined pKa value) CALL CALC_HEFF( pKa, pH, KH, HEFF, RC ) ! Use Henry's Law to get the ratio: ! [ mixing ratio in liquid phase / mixing ratio in gas phase ] L2G = HEFF * H2OLIQ
where:
- K0 (formerly KStar298) is the Henry's law solubility constant [M atm-1]
- CR (formerly H_298) is the Henry's law volatility constant [K]
- pKa is the pH correction factor for the given species [1]
- TK_8 is the temperature [K], in 8-byte floating point
- KH is the dimensionless Henry's law liquid/gas constant for a given species at temperature TK
- HEFF is KH, adjusted for pH. If the species is not pH-sensitive, then HEFF = KH.
- H2OLIQ is the liquid water content [cm3 H2O cm-3 air]
As implemented, the new algorithm is equivalent to the old algorithm except for some minor differences in the definition of physical constants:
- Reference temperature of 298.15 K (new) vs. 298K (old)
- The gas constant R in the new algorithm is defined to more decimal places than in the old algorithm.
- The quantity R / ATM in the new algorithm is equivalent to R in the old algorithm.
- Routines CALC_KH and CALC_HEFF use 8-byte floating point variables instead of flexible-precision. This is to ensure numerical stability in the EXP() calls.
Versions of GEOS-Chem using this new algorithm for computing L2G will contain differences at the level of numerical noise (i.e. absolute differences of order 10-7 when compared to older GEOS-Chem versions.
NOTE: Both the old and new algorithms assume a pH value of 4.5 for rainwater. At present none of the GEOS-Chem species use a correction for pH but this may be changed in the future.
These modifications will be introduced in GEOS-Chem v11-01e.
--Bob Yantosca (talk) 21:40, 15 December 2015 (UTC)
Resolve very high tracer concentrations in MERRA and GEOS-FP wet deposition
This update was validated with 1-month benchmark simulation v11-01d and 1-year benchmark simulation v11-01d-Run1. This version was approved on 12 Dec 2015.
NOTE: This post, which provides a fix for an issue first noticed in the algorithm for convective scavenging of soluble tracers, has been moved to our Cloud convection wiki page.
--Bob Yantosca (talk) 20:15, 10 February 2016 (UTC)
Bugs in MERRA wet deposition
These fixes were included in the GEOS-Chem v10-01 public release (17 Jun 2015).
Carey Friedman wrote:
- I think I've found two bugs in the MERRA wet deposition code that potentially affect all MERRA users but have particular relevance for Hg/POPs users.
- In the POPs model, which I am currently developing (i.e., not the public version), wet deposition mass is passed to depo_pops_mod.F via the routines ADD_POPG_WD and ADD_POPP_WD in convection_mod.F and wetscav_mod.F. This is analogous to what happens in the mercury model: wet dep mass is passed to depo_mercury_mod.F via ADD_Hg2_WD and ADD_HgP_WD. For POPs (and I suspect for Hg, since I literally copied the Hg code, though I haven't tested Hg explicitly), the mass that is passed to the depo module is less than the mass that is archived in the wet dep diagnostics (AD38 and AD39) when MERRA is used. They are equal using GEOS5. For Hg, I think this means mass is lost going from the air to the ocean.
- I've found two areas in the MERRA wet dep code that seem to be causing mass to be lost. The first, in convection_mod.F, is in the DO_MERRA_CONVECTION subroutine for washout of non-aerosol tracers below the cloud base. The current code looks like this:
! MASS_NOWASH is the amount of non-aerosol tracer in
! grid box (I,J,L) that is NOT available for washout.
MASS_NOWASH = ( 1d0 - F_WASHOUT ) * Q(K,IC)
! MASS_WASH is the total amount of non-aerosol tracer
! that is available for washout in grid box (I,J,L).
! It consists of the mass in the precipitating
! part of box (I,J,L), plus the previously rained-out
! tracer coming down from grid box (I,J,L+1).
! (Eq. 15, Jacob et al, 2000).
MASS_WASH = ( F_WASHOUT * Q(K,IC) ) + T0_SUM
! WETLOSS is the amount of tracer mass in
! grid box (I,J,L) that is lost to washout.
! (Eq. 16, Jacob et al, 2000)
WETLOSS = MASS_WASH * WASHFRAC - T0_SUM
- When I change the last equation to
WETLOSS = (MASS_WASH - T0_SUM)* WASHFRAC
- then the mass passed to depo_pops_mod.F and the AD38 diagnostic become equal. This seems to make sense physically based on the comments, but I'm hoping someone more familiar with the convection process can confirm this. If this indeed fixes a bug, then there is analogous code for washout of non-aerosol tracers in DO_WASHOUT_ONLY in wetscav_mod.F that needs to be changed too.
- The second, in wetscav_mod.F, is in the DO_WASHOUT_ONLY subroutine. The following lines, when commented, allow the mass passed to depo_pops_mod.F and AD39 to be equal:
#if defined( MERRA ) || defined( GEOS_57 )
!%%% BUG FIX: Only apply this section of when running
!%%% GEOS-Chem with MERRA met fields. (bmy, hamos, 5/26/11)
!
! Check if WASHFRAC = NaN or WASHFRAC < 0.1 %
!
! If WASHFRAC = NaN, then DSTT = NaN and SAFETY trips because
! tracer concentrations must be finite.WASHFRAC = NaN when F = 0.
! When less than 0.1% of a soluble tracer is available for washout
! DSTT < 0 and SAFETY trips. (Helen Amos, 20100928)
IF ( IT_IS_NAN( WASHFRAC ) ) THEN
CYCLE
ELSEIF ( WASHFRAC < 1D-3 ) THEN
CYCLE
ENDIF
#endif
- If I change this code to
IF ( IT_IS_NAN( WASHFRAC ) ) THEN
CYCLE
ELSEIF ( WASHFRAC < 1D-20 ) THEN
CYCLE
ENDIF
- rather than commenting it, then this reduces the amount of mass that is lost, but the % lost slowly increases over time such that is becomes significant over the course of several years. This is a problem for POPs, some of which run for >100 years.
- Incidentally, POPs also passes dry dep to depo_pops_mod.F the same way Hg does and there are no problems there going between GEOS5 and MERRA.
Helen Amos wrote:
- Remembering back 4-5 years ago when we did this... the fix is only needed for MERRA because the problem was never encountered with GEOS-5 during testing. We never figured out why MERRA met fields had the problem and GEOS-5 didn't, other than the problem seemed to be on the data assimilation side. 1D-3 was selected as a lower limit based on a series of tests on what trapped negative DSTT values and what didn't. This isn't pretty or exact, so if you come up with a better solution, I definitely welcome it!
- DSTT was tested with the Hg and Pb-Be-Rn simulations.
Carey Friedman wrote:
- I would recommend further investigation, since as Helen said, at one point 1d-3 was deemed the lowest cutoff before you started tripping SAFETY. So I don't want to recommend 1d-20, or to comment the code (even though it seems to work for POPs) only to cause people's runs to crash.
- My best recommendation would be to first fix the multiplication by WASHFRAC issue in DO_MERRA_CONVECTION (convection_mod.F) and DO_WASHOUT_ONLY (wetscav_mod.F) and then re-test Hg and Pb-Be-Rn to see if the trap to keep SAFETY from tripping is still needed at all. The WASHFRAC multiplication bugs will make WETLOSS less than it should be, and that could be contributing to the DSTT < 0 problem.
--Melissa Sulprizio 15:37, 2 February 2015 (EST)
Viral Shah wrote:
- I have been also examining these bugs in the wetscav and convection routines, and looking at how they affect the Hg simulation, in particular. My findings/recommendations are as follows:
- 1) For the WASHOUT_ONLY routine (in WETSCAV_MOD):
- The condition requiring that the WASHFRAC>1D-3 or any number greater than 0 for washout to occur, prevents the partial resuspension of the dissolved mass falling from above, and leads to an overestimate in the wet deposited mass. This is consistent with Shaojie's results of an increase in the atmospheric mass of Hg0 following this fix. This also leads to a lower Hg2 mass in the lower troposphere. I recommend that the condition on WASHFRAC should be removed.
- 2) For the DO_MERRA_CONVECTION routine (CONVECTION_MOD):
- I think, the bug here is very similar to the first bug. A condition on F(K,IC)>0 prevents the archiving of deposited mass in the wet deposition diagnostic, but it is recorded in the mass passed from the air to the ocean. After comparing with Jacob et al (2000), I think the current code for calculating washout follows the documented algorithm well, and does not seem to be causing the bug. It would be great to see if this fixes the issue in Carey's POP simulation.
- 3) In the course of reviewing the code, I found two other bugs in DO_MERRA_CONVECTION. First, the code is missing a statement that subtracts the wet deposited amount from the atmospheric mass. And, second, there seems to be an inconsistency in units with how the variable T0_SUM was used.
- I have attached a document describing this is more detail, and the fixes I propose for these bugs. I am also attaching the following link to the benchmark plots, comparing the results with the original code and the code with my proposed fixes.
--Melissa Sulprizio (talk) 20:30, 20 May 2015 (UTC)
Bug fixes for scavenging by co-condensation
These updates were validated with the 1-month benchmark simulation v10-01f and approved on Approved 13 Jan 2015.
NOTE: The PARAMETER statements below were removed from wetdep_mod.F in v11-01d. These values are now specified via the species database field WD_ConvFacI2G.
Duncan Fairlie wrote:
- One detail that has got me puzzled concerns scavenging by co-condensation:
- Formulation under Mari et al. (2000) (for H2O2) for fraction in solid phase looks like
- Whereas in the code wetdep scheme it looks like
- Where W is the cloud LWC. I.e. The square root term is inverted in the 2 formulations.
- The code changes apply to subroutine COMPUTE_F, and concern the parameters CONV_H2O2 and CONV_NH3. To be consistent with the formulation in Mari et al. (2000), CONV_H2O2 for example should be defined as
- And similarly for CONV_NH3. I.e. The square-root term should be inverted w.r.t. what is currently in the code.
- Replace lines:
! CONV_H2O2 = 0.6 * SQRT( 1.9 ), used for the ice to gas ratio for H2O2
! 0.6 is ( sticking coeff H2O2 / sticking coeff water )
! 1.9 is ( molecular weight H2O2 / molecular weight water )
REAL*8, PARAMETER :: CONV_H2O2 = 8.27042925126d-1
! CONV_NH3 = 0.6 * SQRT( 0.9 ), used for the ice to gas ratio for NH3
! 0.6 is ( sticking coeff NH3 / sticking coeff water )
! 0.9 is ( molecular weight NH3 / molecular weight water )
REAL*8, PARAMETER :: CONV_NH3 = 5.69209978831d-1
- With
! CONV_H2O2 = 0.6 * SQRT( 0.53 ), used for the ice to gas ratio for H2O2
! 0.6 is ( sticking coeff H2O2 / sticking coeff water )
! 0.53 is ( molecular weight water / molecular weight H2O2 )
REAL*8, PARAMETER :: CONV_H2O2 = 4.36564d-1
! CONV_NH3 = 0.6 * SQRT( 1.06 ), used for the ice to gas ratio for NH3
! 0.6 is ( sticking coeff NH3 / sticking coeff water )
! 1.06 is ( molecular weight water / molecular weight NH3 )
REAL*8, PARAMETER :: CONV_NH3 = 6.17395d-1
- That should correct the issue. These changes should also apply to the RAINOUT subroutine, in addition to COMPUTE_F.
- This also applies to Equ. 9 in the documentation of wet scavenging: Jacob et al., 2000, Harvard wet deposition scheme for GMI. The term sqrt(MH2O2/MH2O) in Equ. 9 should read sqrt(MH2O/MH2O2). With associated changes to the text following.
--Melissa Sulprizio 14:56, 19 December 2014 (EST)
Prevent div-by-zero in WASHFRAC routines
These fixes were tested with 1-month benchmark GEOS-Chem v9-02p and approved on 13 Sep 2013.
In the following routines in GeosCore/wetscav_mod.F:
- WASHFRAC_FINE_AEROSOL
- WASHFRAC_COARSE_AEROSOL
- WASHFRAC_SIZE_AEROSOL
- WASHFRAC_HNO3
we now make sure to only compute the WASHFRAC quantity when F > 0. In other words, wherever we see an expression of the form:
WASHFRAC = F *(1d0 - EXP( -K_WASH *
& (PP / F*3.6d4 )**0.61d0 * DT / 3.6d3 ))
we now bracket this with an IF statement, such as this:
IF ( F > 0d0 ) THEN
WASHFRAC = F *(1d0 - EXP( -K_WASH *
& (PP / F*3.6d4 )**0.61d0 * DT / 3.6d3 ))
ELSE
WASHFRAC = 0d0
ENDIF
Because F multiplies the entire expression, WASHFRAC would become zero whenever F=0. But the IF statement also avoids placing F=0 into the denominator of a division, which would result in a div-by-zero condition.
This error condition was caught by the GEOS-Chem unit tester when compiling with option FPE=yes.
--Bob Y. 12:19, 12 September 2013 (EDT)
Washout fix for non-aerosol species
A detailed description of this fix can be found in the Appendix of Amos et al. (2012, ACP).
This update was tested in the 1-month benchmark simulation v9-01-02f and approved on 02 Aug 2011.
Prior to GEOS-Chem v9-01-02, this bug was causing the wet deposition algorithm to underestimate washout of highly soluble gases:
Helen Amos wrote:
- In section 2.2.2 of Jacob et al. (2000), the first bullet point states:
If fi,L > Fi/f, scavenging is limited by mass transfer and we follow exactly the same procedure as for aerosols and HNO3 (section 2.2.1).
- Prior to GEOS-Chem v9-01-02, WASHFRAC_LIQ_GAS was not executing washout of non-aerosol tracers as dictated by this statement.
- Washout proceeds in two ways: (1) Henry's law equilibrium and (2) limited by mass transfer. Aerosols, HNO3, and highly soluble gases should proceed through washout obeying the mass transfer limitation. Moderately and sparingly soluble gases should proceed through washout obeying Henry's law equilibrium. The purpose of WASHFRAC_LIQ_GAS is to calculate WASHFRAC for non-aerosol species, where WASHFRAC is the fraction of a tracer available for washout. WASHFRAC is calculated using Henry's law if fi,L <= Fi/f (Eqn 15-16, Jacob et al. 2000), and WASHFRAC is calculated using mass transfer if fi,L > Fi/f (Eqn 14, Jacob et al. 2000). The algorithms in WASHOUT are written such that if you specify that a tracer proceed through washout according to Henry's law, the code expects WASHFRAC to be calculated using Henry's law (Eqn 15-16, Jacob et al. 2000). Similarly, if you specify that a tracer proceed through washout limited by mass transfer, then the code expects WASHFRAC to be calculated using mass transfer (Eqn 14). Prior to GEOS-Chem v9-01-02, every soluble gas (except HNO3) was arbitrarily forced to proceed through washout according to Henry's law, even when fi,L > Fi/f and WASHFRAC had been calculated from mass transfer equations. In short, there was a mismatch in information between WASHFRAC_LIQ_GAS and WASHOUT. This mismatch caused an underestimate in the washout of highly soluble gases because too much tracer was being re-evaporated (negative term in WETLOSS, where WETLOSS is the amount of tracer lost in a grid box).
- A logical is now passed as an input/output parameter to WASHFRAC_LIQ_GAS and this logical will specify whether a non-aerosol species should proceed through washout according to Henry's law equilibrium or limited by mass transfer. The code is now consistent with Section 2.2.2 of Jacob et al. (2000).
This fix was originally tested in the 1-month benchmark simulation v9-01-02b. A second bug in WASHFRAC_LIQ_GAS was discovered, invalidating v9-01-02b (see discussion below). The complete fix for WASHFRAC_LIQ_GAS has been added to the standard code and approved in the 1-month benchmark simulation v9-01-02f.
Update 7/28/2011: The original algorithm we tested in 1-month benchmark v9-01-02b was incorrect. Prior to v9-01-02b, routine WASHFRAC_LIQ_GAS looked like this:
IF (WASHFRAC > WASHFRAC_F_14) THEN WASHFRAC = WASHFRAC_F_14
In benchmark simulation v9-01-02b we had modified this IF statement (incorrectly) to:
IF ( WASHFRAC > WASHFRAC_F_14 ) THEN WASHFRAC = WASHFRAC_F_14 AER = TRUE. ENDIF
However, this IF statement should have been:
IF ( WASHFRAC > WASHFRAC_F_14 ) THEN WASHFRAC = F * WASHFRAC_F_14 AER = TRUE. ENDIF
This fix has been added to the standard code and was approved in the v9-01-02f benchmark. For more information, please refer to the Appendix of Amos et al. (2012, ACP).
--Helen Amos 03:57, 13 Aug 2011 (EST)
Outstanding issues
Low tropospheric 210Pb lifetime against deposition in v11-01b
Due to the high impact on aerosols, the GEOS-Chem Steering Committee rejected the quick fix listed below. We have subsequently removed this fix from v11-01d and performed additional 1-year benchmark simulations with the updated model. The retuning of wet deposition for GEOS-FP and MERRA-2 met fields is still an active area of reaearch.
Hongyu Liu wrote:
- The tropospheric 210Pb lifetime against deposition of 6.6 days (G-C/GEOS-FP) is the shortest among G-C versions. Switching from GEOS-5 to GEOS-FP met fields (v9-02r) reduced 210Pb lifetime from 9.3 to 7.7 days. See: http://wiki.seas.harvard.edu/geos-chem/index.php/Rn-Pb-Be_simulation#Budget_of_Pb210. If v11-01b had been driven by GEOS-5, the 210Pb lifetime may very well be ~8 days. Wang, Q. et al. [2014] used G-C/GEOS-5 and "find a lifetime of tropospheric 210Pb aerosol against deposition of 8.6 days, as compared to a best estimate of 9 days constrained by observations [Liu et al., 2001]." So it appears that the short 210Pb lifetime (6.6 days) in v11-01b is largely due to switching to GEOS-FP. Wet deposition needs to be re-evaluated and tuned for GEOS-FP.
- Bo Zhang has done a series of G-C v11-01b simulations of 210Pb with GEOS-FP (2013 - year for benchmark) and GEOS-5 (2009 - year used in Wang et al. 2014)...We were able to reproduce the 210Pb lifetime (GEOS-5, 2009) reported in Wang Q. et al. (2014) using the standard v11-01b code, where scavenging due to cold cloud is not applied to 210Pb. Switching from GEOS-5 to FP reduces 210Pb lifetime by about 2 days. (But note that G-C/GEOS5 uses MOISTQ to approximate 3-D precipitation formation rates while G-C/FP takes the precipitation formation rates from the FP archive.) Bo is currently evaluating these simulations with 210Pb surface concentration / flux & UT/LS observations.
Bo Zhang and Hongyu Liu provided the following quick fix recommendation on 10/8/15 that was initially implemented in v11-01d but then removed pending further testing:
- Recommendation for a quick fix: For GEOS-FP (and MERRA-2), turn off cold cloud scavenging & change the condensed water content (CWC) back to 1.5x10-3kg/m3. The resulting 210Pb aerosol RT against wetdep (7.8 days) would be close to that in GEOS-5 (with cold cloud scaveging & CWC=1x10-3kg/m3, Q. Wang et al. 2014). This is to be implemented as follows:
- For GEOS-5 and MERRA:
- Keep what the original code (Q. Wang et al.) was doing.
- Add cold cloud scavenging to 210Pb-7Be (when T < 258 K).
- For GEOS-FP and MERRA-2:
- For ALL aerosols (including 210Pb-7Be):
- Turn off the cold cloud scavenging (i.e. set RAINFRAC = 0) when T < 258 K.
- Without this tuning, the scavenging in GEOS-FP and MERRA-2 would be too fast.
- For ALL aerosols (including 210Pb-7Be):
The tables below summarize model results for v11-01d with and without the quick fix described above.
Pb210 and Be7 budgets
| Version | Met Field | Year | Tropospheric burden [g] | Tropospheric lifetime against deposition [days] | Sources [g day -1] | Sinks [g day-1] | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| From Stratosphere | From Troposphere | Dry Deposition | Wet Deposition | Radioactive decay | |||||||
| Total | Stratiform | Convective | |||||||||
| Pb210 | |||||||||||
| v11-01d w/o quick fix |
GEOS-FP | 2013 | 210.371 | 6.54296 | 0.225956 | 31.9538 | 3.41587 | 28.7451 | 21.9070 | 6.83813 | 0.0177696 |
| v11-01d w/ quick fix |
GEOS-FP | 2013 | 281.906 | 8.76224 | 0.244503 | 31.9528 | 4.07200 | 28.1015 | 19.4702 | 8.63134 | 0.0237767 |
| Be7 | |||||||||||
| v11-01d w/o quick fix |
GEOS-FP | 2013 | 3.32564 | 23.2523 | 0.0530363 | 0.132914 | 0.00664940 | 0.135989 | 0.122347 | 0.0136424 | 0.0433123 |
| v11-01d w/ quick fix |
GEOS-FP | 2013 | 4.15303 | 31.7653 | 0.0516775 | 0.132914 | 0.01025760 | 0.120259 | 0.0974579 | 0.0228011 | 0.0540753 |
Global Tropospheric Aerosol Burdens
| Version | Met Field | Year | BCPI [Tg] | SO4 [Tg] | DST1 [Tg] | SALA [Tg] | SALC [Tg] |
|---|---|---|---|---|---|---|---|
| v11-01d w/o quick fix |
GEOS-FP | 2013 | 0.0643855 | 1.16159 | 1.73150 | 0.305502 | 3.41605 |
| v11-01d w/ quick fix |
GEOS-FP | 2013 | 0.08122 | 1.61810 | 2.13533 | 0.39143 | 3.76399 |
Global Tropospheric Mean Aerosol Optical Depth
| Version | Met Field | Year | Dust (OPD) | Sulfate (OPSO4550) | Black Carbon (OPBC550) | Organic Carbon (OPOC550) | Sea Salt (accum) (OPSSa550) | Sea Salt (coarse) (OPSSc550) |
|---|---|---|---|---|---|---|---|---|
| v11-01d w/o quick fix |
GEOS-FP | 2013 | 0.0097336 | 0.0256808 | 0.0007353 | 0.0093898 | 0.0137217 | 0.0277927 |
| v11-01d w/ quick fix |
GEOS-FP | 2013 | 0.0112812 | 0.0339483 | 0.0008956 | 0.0114021 | 0.0168340 | 0.0308662 |
In response to the v11-01d 1-month and 1-year benchmark results, Peter Adams writes:
- Colette and I have been through the benchmarks and just got off the phone to discuss. We agreed on the following:
- The “quick fix” implemented by Hongyu to wet scavenging has significant effects on aerosols species. Most notably, free tropospheric levels of sulfate and black carbon increase by a factor of ~2. Changes in overall burdens are less, of course, but still significant.
- With a change of this magnitude for aerosols, we would much rather proceed with something that has undergone some substantial vetting. Based on our communications with Hongyu, this is very much a “quick fix” – one that his group will be evaluating in much more detail in the next months. We all know that wet deposition lifetimes are the result of a bunch of contributing processes. As far as I can tell, we are not sure that making a change specifically to cold cloud scavenging pushes us in the right direction for the right reasons.
- For these reasons, both Colette and I recommend removing – at least for now – Hongyu’s cold cloud fix until it gets more evaluation. To be clear, this is more for the sake of making significant changes to physics only after careful evaluation rather than clear-cut observational evidence that model predictions are better/worse with or without the fix.
--Melissa Sulprizio (talk) 16:44, 4 December 2015 (UTC)
References
- Amos, H. M., D. J. Jacob, C. D. Holmes, J. A. Fisher, Q. Wang, R. M. Yantosca, E. S. Corbitt, E. Galarneau, A. P. Rutter, M. S. Gustin, A. Steffen, J. J. Schauer, J. A. Graydon, V. L. St. Louis, R. W. Talbot, E. S. Edgerton, Y. Zhang, and E. M. Sunderland, Gas-Particle Partitioning of Atmopsheric Hg(II) and Its Effect on Global Mercury Deposition, Atmos. Chem. Phys.,12,591-603, 2012. PDF
- Dana, M.T., and J.M. Hales, Statistical aspects of the washout of polydisperse aerosols, Atmos. Environ, 10, 45-50, 1976.
- Domine, F., and E. Thibert, Mechanism of incorporation of trace gases in ice grown from the gas phase, Geophys. Res. Lett., 23, 3627-3630, 1996.
- Giorgi, F., and W.L. Chameides, Rainout lifetimes of highly soluble aerosols as inferred from simulations with a general circulation model, J. Geophys. Res., 91, 14,367-14,376, 1986.
- Jacob, D.J., Heterogeneous chemistry and tropospheric ozone, Atmos. Environ., 34, 2131-2159, 2000. PDF
- Jacob, D.J. H. Liu, C.Mari, and R.M. Yantosca, Harvard wet deposition scheme for GMI, Harvard University Atmospheric Chemistry Modeling Group, revised March 2000. PDF
- Levine, S.Z., and S.E. Schwartz, In-cloud and below-cloud scavenging of nitric acid vapor, Atmos. Environ., 16, 1725-1734, 1982.
- Liu, H., D.J. Jacob, I. Bey, and R.M. Yantosca, Constraints from 210Pb and 7Be on wet deposition and transport in a global three-dimensional chemical tracer model driven by assimilated meteorological fields, J. Geophys. Res., 106, 12,109-12,128, 2001. PDF
- Mari, C., D.J. Jacob, and P. Bechtold, Transport and scavenging of soluble gases in a deep convective cloud, J. Geophys. Res., 105, 22,255-22,267, 2000. PDF
- Selin, N.E. and D.J. Jacob, Seasonal and spatial patterns of mercury wet deposition in the United States: North American vs. intercontinental sources, Atmospheric Environment, 42, 5193-5204, 2008. PDF
- Wang, Q., D.J. Jacob, J.A. Fisher, J. Mao, E.M. Leibensperger, C.C. Carouge, P. Le Sager, Y. Kondo, J.L. Jimenez, M.J. Cubison, and S.J. Doherty, Sources of carbonaceous aerosols and deposited black carbon in the Arctic in winter-spring: implications for radiative forcing, Atmos. Chem. Phys., 11, 12,453-12,473, 2011. PDF
- Wang, Q., D.J. Jacob,J.R Spackman, A.E. Perring, J.P. Schwarz, N. Moteki, E.A. Marais, C. Ge, J. Wang and S.R.H. Barrett, Global budget and radiative forcing of black carbon aerosol: constraints from pole-to-pole (HIPPO) observations across the Pacific, J. Geophys. Res., 119, 195-206, 2014. PDF