Difference between revisions of "Caltech isoprene scheme"
(→New reactions) |
(→New reactions) |
||
| Line 302: | Line 302: | ||
|47||IEPOX + OH = IEPOXOO ||5.78e-11exp(-400/T) ||Paulot 2009b || | |47||IEPOX + OH = IEPOXOO ||5.78e-11exp(-400/T) ||Paulot 2009b || | ||
|- | |- | ||
| − | |48||IEPOXOO + HO2 = 0.725HAC + 0.275GLYC + 0.275GLYX + 0.275MGLY + 1.125OH + 0.825HO2 + 0.200CO2 + 0.375CH2O + 0.074HCOOH + 0.251CO || | + | |48||IEPOXOO + HO2 = 0.725HAC + 0.275GLYC + 0.275GLYX + 0.275MGLY + 1.125OH + 0.825HO2 + 0.200CO2 + 0.375CH2O + 0.074HCOOH + 0.251CO ||2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 ||Paulot 2009b || |
|- | |- | ||
|49||IEPOXOO +NO=0.725HAC+0.275GLYC+0.275GLYX +0.275MGLY +0.125OH +0.825HO2+0.200CO2+0.375CH2O +0.074HCOOH +0.251CO +NO2 ||2.7E-12exp(350/T) || ||FP: No peroxide was observed | |49||IEPOXOO +NO=0.725HAC+0.275GLYC+0.275GLYX +0.275MGLY +0.125OH +0.825HO2+0.200CO2+0.375CH2O +0.074HCOOH +0.251CO +NO2 ||2.7E-12exp(350/T) || ||FP: No peroxide was observed | ||
| Line 316: | Line 316: | ||
|54||MAOP + OH = MAO3 ||6.13E-13exp(200/T) || ||same as MAP+OH | |54||MAOP + OH = MAO3 ||6.13E-13exp(200/T) || ||same as MAP+OH | ||
|- | |- | ||
| − | |55||MAOP+OH=MAOPO2 ||3.60E-12exp(380/T) || || | + | |55||MAOP + OH = MAOPO2 ||3.60E-12exp(380/T) || || |
|- | |- | ||
| − | |56||MCO3+MAOPO2=1.0HAC+2.0CO2+OH+MO2 ||1.68E-12exp(500/T) || || | + | |56||MCO3 + MAOPO2 = 1.0HAC + 2.0CO2 + OH + MO2 ||1.68E-12exp(500/T) || || |
|- | |- | ||
| − | |57||MCO3+MAOPO2=1.0ACTA+1.0MEK ||1.87E-13 exp(500/T) || || | + | |57||MCO3 + MAOPO2 = 1.0ACTA+1.0MEK ||1.87E-13 exp(500/T) || || |
|- | |- | ||
| − | |58||MAOPO2+MO2=0.7HAC +0.7CO2+0.7OH+1.0CH2O+0.7HO2+0.3ROH ||8.37E-14 || || | + | |58||MAOPO2 + MO2 = 0.7HAC +0.7CO2+0.7OH+1.0CH2O+0.7HO2+0.3ROH ||8.37E-14 || || |
|- | |- | ||
| − | |59||MAOPO2+MAOPO2=2.0HAC+2.0CO2+2.0OH ||8.37E-14 || || | + | |59||MAOPO2 + MAOPO2 = 2.0HAC+2.0CO2+2.0OH ||8.37E-14 || || |
|- | |- | ||
| − | |60||MAOPO2+HO2=1.0HAC+1.0CO2+2.0OH || | + | |60||MAOPO2 + HO2 = 1.0HAC+1.0CO2+2.0OH ||2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=4 || || |
|- | |- | ||
| − | |61||MAOPO2+NO=1.0HAC+1.0CO2+1.0OH+1.0NO2 ||K* (1-YN) where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=4.00E00) || || | + | |61||MAOPO2 + NO = 1.0HAC+1.0CO2+1.0OH+1.0NO2 ||K* (1-YN) where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=4.00E00) || || |
|- | |- | ||
| − | |62||MAOPO2+NO=1. | + | |62||MAOPO2 + NO = 1.0HNO3 ||K* YN where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=4.00E00) || ||Not treated explicitly |
|- | |- | ||
| − | |63||OH+MAP=1.0MCO3 ||6.13E-13exp(200/T) || ||From J. Orlando (unpublished results), how confident is this temperature dependence? | + | |63||OH + MAP = 1.0MCO3 ||6.13E-13exp(200/T) || ||From J. Orlando (unpublished results), how confident is this temperature dependence? |
|- | |- | ||
| − | |64||HC5+O3 = 0.6MGLY + 0.1OH + 0.12CH2O + 0.28GLYC + 0.3O3 + 0.4CO + 0.2H2 + 0.2HAC + 0.2HCOOH ||6.16E-15 exp(-1814/T)|| ||HC5=old IALD?? | + | |64||HC5 + O3 = 0.6MGLY + 0.1OH + 0.12CH2O + 0.28GLYC + 0.3O3 + 0.4CO + 0.2H2 + 0.2HAC + 0.2HCOOH ||6.16E-15 exp(-1814/T)|| ||HC5=old IALD?? |
|- | |- | ||
| − | |65||R4O2+MCO3 = MO2 +0.32ACET+0.19MEK+0.27HO2+0.32ALD2+0.13RCHO+0.05A3O2+0.18B3O2+0.32ETO2 ||1.68E-12 exp(500/T) || || | + | |65||R4O2 + MCO3 = MO2 + 0.32ACET + 0.19MEK + 0.27HO2 + 0.32ALD2 + 0.13RCHO + 0.05A3O2 + 0.18B3O2 + 0.32ETO2 ||1.68E-12 exp(500/T) || || |
|- | |- | ||
|66||ATO2+MCO3 = MCO3 +CH2O +MO2 ||1.68E-12 exp(500/T) ||IUPAC06 || | |66||ATO2+MCO3 = MCO3 +CH2O +MO2 ||1.68E-12 exp(500/T) ||IUPAC06 || | ||
| Line 342: | Line 342: | ||
|67||RIO2 + MCO3 = 0.887HO2 + 0.747CH2O + 0.453MVK + 0.294MACR + 0.140HC5 + 0.113DIBOO + CO2 + MO2||1.68E-12 exp(500/T) || ||Follow RIO2+NO without the yield of nitrate and NO2 and then rescale it. | |67||RIO2 + MCO3 = 0.887HO2 + 0.747CH2O + 0.453MVK + 0.294MACR + 0.140HC5 + 0.113DIBOO + CO2 + MO2||1.68E-12 exp(500/T) || ||Follow RIO2+NO without the yield of nitrate and NO2 and then rescale it. | ||
|- | |- | ||
| − | |68||HC5OO +MCO3=0.216GLYX +0.234MGLY+0.234GLYC+0.216HAC +0.29DHMOB+0.17MOBA+0.09RCHO+HO2+0.09CO+MO2 ||1.68E-12 exp(500/T) || ||HC5OO=old IAO2, this radical channel use the same as HC5OO+NO without NO2 yield. | + | |68||HC5OO + MCO3 = 0.216GLYX + 0.234MGLY + 0.234GLYC + 0.216HAC + 0.29DHMOB + 0.17MOBA + 0.09RCHO + HO2 + 0.09CO + MO2 ||1.68E-12 exp(500/T) || ||HC5OO=old IAO2, this radical channel use the same as HC5OO+NO without NO2 yield. |
|- | |- | ||
| − | |69||VRO2+MCO3=0.4HO2+0.4CH2O+0.6MCO3+0.6GLYC+0.4MGLY+1.0MO2 ||1.68E-12 exp(500/T) || ||this radical channel use the same as VRO2+NO without NO2 and MVKN yield. And carbon balance. | + | |69||VRO2 + MCO3 = 0.4HO2 + 0.4CH2O + 0.6MCO3 + 0.6GLYC + 0.4MGLY + 1.0MO2 ||1.68E-12 exp(500/T) || ||this radical channel use the same as VRO2+NO without NO2 and MVKN yield. And carbon balance. |
|- | |- | ||
| − | |70||MRO2 +MCO3 | + | |70||MRO2 + MCO3 = 0.850HO2 + 0.143MGLY + 0.857HAC + 0.857CO + 0.143CH2O +1.0MO2 ||1.68E-12 exp(500/T) || ||this radical channel use the same as MRO2+NO without NO2 and MACRN yield. |
|- | |- | ||
|71||MAN2+MCO3 = MO2 +.5NO2+.5CH2O+.5MGLY+.5PROPNN+.5CO+.5HO2 ||1.68E-12 exp(500/T) || || | |71||MAN2+MCO3 = MO2 +.5NO2+.5CH2O+.5MGLY+.5PROPNN+.5CO+.5HO2 ||1.68E-12 exp(500/T) || || | ||
| Line 354: | Line 354: | ||
|73||HC5OO+MCO3 = MEK +ACTA ||1.87E-13 exp(500/T) || || | |73||HC5OO+MCO3 = MEK +ACTA ||1.87E-13 exp(500/T) || || | ||
|- | |- | ||
| − | |74||ISOPNB+O3=0.610MVKN +0.390MACRN +0. | + | |74||ISOPNB + O3 = 0.610MVKN + 0.390MACRN + 0.27OH + CH2O ||1.06E-16 ||Lockwood et al., 2010 ACP ||use 1,2 for beta channel |
|- | |- | ||
| − | |75||ISOPND+O3=0. | + | |75||ISOPND + O3 = 0.5PROPNN + 0.5ETHLN + 0.27OH + 0.5GLYC + 0.5HAC ||5.3E-17 ||Lockwood et al., 2010 ACP ||use 1,4 for delta channel |
|- | |- | ||
|76||HC5+OH=HC5OO ||3.35E-11exp(380/T) ||Paulot 2009a || | |76||HC5+OH=HC5OO ||3.35E-11exp(380/T) ||Paulot 2009a || | ||
| Line 364: | Line 364: | ||
|78||HC5OO +NO=HNO3 ||K* YN where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=5.00E00) || || | |78||HC5OO +NO=HNO3 ||K* YN where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=5.00E00) || || | ||
|- | |- | ||
| − | |79||HC5OO +HO2 =0.1IAP+0.9OH +0.9MGLY | + | |79||HC5OO + HO2 = 0.1IAP + 0.9OH + 0.9MGLY + 0.9GLYC + 0.9HO2 ||2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 || ||90% recycling, no experimental data. Somewhat based upon the high recycling rate observed for MVK/MACR |
|- | |- | ||
|80||ISOPND+OH=ISOPNDO2 ||2.64E-11exp(380/T) ||Paulot 2009a|| | |80||ISOPND+OH=ISOPNDO2 ||2.64E-11exp(380/T) ||Paulot 2009a|| | ||
| Line 374: | Line 374: | ||
|83||ISOPNDO2+NO=HNO3 ||K* YN where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=5.00E00) || ||Nitrates from ISOPND could not be observed in this experiment | |83||ISOPNDO2+NO=HNO3 ||K* YN where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=5.00E00) || ||Nitrates from ISOPND could not be observed in this experiment | ||
|- | |- | ||
| − | |84||ISOPNDO2 + HO2 = 0.035MACRN + 0.155HCOOH + 0.22HAC + 0.065ETHLN + 0.325CH2O + 0.170NO2 + 0.075GLYC + 0.155NO3 + 0.075PROPNN + 0.170MEK + 0.175HO2 + 0.5OH + 0.5ISNP || | + | |84||ISOPNDO2 + HO2 = 0.035MACRN + 0.155HCOOH + 0.22HAC + 0.065ETHLN + 0.325CH2O + 0.170NO2 + 0.075GLYC + 0.155NO3 + 0.075PROPNN + 0.170MEK + 0.175HO2 + 0.5OH + 0.5ISNP ||2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 || ||Assume 50% recycling from HO2+RO2 (no experimental data - check Ng et al. for better estimates) |
|- | |- | ||
|85||ISOPNBO2 + NO = 0.6GLYC + 0.6HAC + 0.4CH2O + 1.6NO2 + 0.26MACRN + 0.4HO2 + 0.14MVKN ||K* (1-YN) where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=4.00E00) ||Paulot 2009a || | |85||ISOPNBO2 + NO = 0.6GLYC + 0.6HAC + 0.4CH2O + 1.6NO2 + 0.26MACRN + 0.4HO2 + 0.14MVKN ||K* (1-YN) where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=4.00E00) ||Paulot 2009a || | ||
| Line 380: | Line 380: | ||
|86||ISOPNBO2 + NO = HNO3 ||K* YN where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=5.00E00) || ||Nitrates from ISOPND could not be observed in this experiment | |86||ISOPNBO2 + NO = HNO3 ||K* YN where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=5.00E00) || ||Nitrates from ISOPND could not be observed in this experiment | ||
|- | |- | ||
| − | |87||ISOPNBO2 + HO2 = 0.3GLYC + 0.3HAC + 0.2CH2O + 0.13MACRN + 0.07MVKN + 0.3NO2 + 0.2HO2 + 0.5OH + 0.5ISNP || | + | |87||ISOPNBO2 + HO2 = 0.3GLYC + 0.3HAC + 0.2CH2O + 0.13MACRN + 0.07MVKN + 0.3NO2 + 0.2HO2 + 0.5OH + 0.5ISNP ||2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 || ||Assume 50% recycling from HO2+RO2 (no experimental data - check Ng et al. for better estimates) |
|- | |- | ||
|88||ISNP+OH=0.612OH+0.612R4N1++0.193ISOPNBO2+0.193ISOPNDO2 ||4.75E-12exp(200/T) || ||replace the old ISNP+OH | |88||ISNP+OH=0.612OH+0.612R4N1++0.193ISOPNBO2+0.193ISOPNDO2 ||4.75E-12exp(200/T) || ||replace the old ISNP+OH | ||
| Line 390: | Line 390: | ||
|91||MACRNO2 + NO = 0.08ACTA + 0.08CH2O + 0.15NO3 + 0.07HCOOH + 0.070MGLY + 0.850HAC + 0.85NO2 + 0.93CO2 + 1.0NO2 ||2.7E-12exp(350/T) || ||no nitrate yield (acyl) | |91||MACRNO2 + NO = 0.08ACTA + 0.08CH2O + 0.15NO3 + 0.07HCOOH + 0.070MGLY + 0.850HAC + 0.85NO2 + 0.93CO2 + 1.0NO2 ||2.7E-12exp(350/T) || ||no nitrate yield (acyl) | ||
|- | |- | ||
| − | |92||MACRNO2 + HO2 = 0.08ACTA + 0.08CH2O + 0.15NO3 + 0.07HCOOH + 0.07MGLY + 0.85HAC + 0.85NO2 + 0.93CO2 + 1.0OH || | + | |92||MACRNO2 + HO2 = 0.08ACTA + 0.08CH2O + 0.15NO3 + 0.07HCOOH + 0.07MGLY + 0.85HAC + 0.85NO2 + 0.93CO2 + 1.0OH ||2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=4 || ||Assume 100% recycling. No experiment data. Inferred from the very high recycling observed for MAO2+HO2 |
|- | |- | ||
|93||MACRNO2 + NO2=1.0PMNN ||LPL: 9.00E-28(300/T)^8.9 HPL:7.70E-12(300/T)^0.2 Fc: 0.6 || || | |93||MACRNO2 + NO2=1.0PMNN ||LPL: 9.00E-28(300/T)^8.9 HPL:7.70E-12(300/T)^0.2 Fc: 0.6 || || | ||
Revision as of 17:08, 5 February 2013
NOTE: This page is for documentation of new isoprene chemistry to be included in v9-02.
'Previous page of description can be found here New isoprene scheme prelim.
Contents
Implementation of the Paulot isoprene scheme
A full reference of all reactions used can be found in this document.
Species
New species added
| Species | Formula | Note |
| MAOPO2 | CH2OH-CHOO*CH3C(O)OOH | Peroxy radical from MAOP (addition on the double bond) |
| ISOPNB | C5H9NO4 | Isoprene nitrate Beta |
| ISOPND | C5H9NO4 | Isoprene nitrate Delta |
| HC5 | C55H8O2 | Hydroxycarbonyl with 5C |
| DIBOO | Dibble peroxy radical | |
| MF3 | Methyl furan but I didn’t use it | |
| HC5OO | Peroxy radical from HC5 (old IAO2?) | |
| DHMOB | C5H8O4 | See Paulot et al., ACP (2009) |
| MOBA | 5C acid from isoprene | |
| MOBAOO | RO2 from MOBA | |
| ISOPNBO2 | RO2 from ISOPND | |
| ISOPNDO2 | RO2 from ISOPND | |
| PROPNN | Propanone nitrate | |
| ETHLN | Ethanal nitrate | |
| DHB | Dihydroxybutanone (but removed) | |
| MACRN | Nitrate from MVK | |
| MVKN | Nitrate from MACR | |
| PYAC | Pyruvic acid | |
| IEPOX | Isoprene epoxide | |
| IEPOXOO | RO2 from IEPOX | |
| PYPAN | Pyruvic acid PAN | |
| PYPO2 | RO2 associated with PYPAN | |
| ATOOH | ATO2 peroxide | |
| DRYPMNN | Dry deposition for the different species | |
| DRYALPH | ||
| DRYLIMO | ||
| DRYISOPND | ||
| DRYISOPNB | ||
| DRYRIP | ||
| DRYIEPOX | ||
| DRYMACRN | ||
| DRYMVKN | ||
| DRYPROPNN | ||
| DRYHCOOH | ||
| DRYACTA | ||
| DRYPYPAN |
Species emitted and deposited
| Species emitted | Species deposited |
| NO | NO2 |
| NO2 | O3 |
| CO | PAN |
| ALK4 | HNO3 |
| ISOP | CH2O |
| ACET | N2O5 |
| PRPE | H2O2 |
| C3H8 | PMN |
| C2H6 | PPN |
| MEK | R4N2 |
| ALD2 | |
| CH2O | PMNN |
| HNO3 | IEPOX |
| O3 | RIP |
| ISOPND | |
| ISOPNB | |
| PROPNN | |
| MACRN | |
| MVKN | |
| HCOOH | |
| ACTA |
Henry's law constant
| Species | Heff(moles L-1 atm-1 ) | ΔH/R (K) | Reference |
| HCOOH | 167,000 (pH = 5) | -6100 | Ito et al., 2007 |
| CH3COOH | 11,400 (pH = 5) | -6300 | Ito et al., 2007 |
| MOBA | 23000 | -6300 | Ito et al., 2007 |
| GLYC | 41000 | -4600 | Ito et al., 2007 |
| GLYX | 360000 | -7200 | Schweitzer et al., 1998 |
| MGLY | 3700 | -7500 | Ito et al., 2007 |
| δ-ISOPN (ISOPND) | 17000 | -9200 | Ito et al., 2007 |
| β-ISOPN (ISOPND) | 17000 | -9200 | Ito et al., 2007 |
| MACRN | 17000 | -9200 | Ito et al., 2007 |
| MVKN | 17000 | -9200 | Ito et al., 2007 |
| PROPNN | 1000 | 0 | R. Sander (NITROOXYACETONE) |
| RIP | 1.7e6 | 0 | Marais et al., 2012 |
| IEPOX | 1.3e8 | 0 | Marais et al., 2012 |
| MAP | 840 (f0 =1, reactive) | -5300 | R. Sander |
| HNO3 | 210000 | -8700 |
Species removed from standard chemistry
| Species | Formula | Note |
| GCO3 | HOCH2C(O)OO | hydroxy peroxyacetyl radical |
| GLCO3 | HCOHC(O)OO | peroxyacyl from GLYX |
| GLP | HCOHC(O)OOH | peroxide from GLCO3 |
| GLPAN | HCOHC(O)OONO2 | peroxyacylnitrate from GLCO3 |
| GP | HOCH2C(O)OOH | peroxide from GCO3 |
| GPAN | HOCH2C(O)OONO2 | peroxyacylnitrate from GCO3 |
| ISN1 | HOCH2C(OO)(CH3)CH(ONO2)CH2OH | RO2 from ISN2 |
| RIO1 | HOCH2C(OO)(CH3)CH=CHOH | RO2 from isoprene oxidation products |
| IAO2 | HOCH2C(CH3)(OO)CH(OH)CHO | RO2 from isoprene oxidation products |
| MVN2 | O2NOCH2CH(OO)C(O)CH3 | RO2 from MVK+NO3 |
Species Activated from standard chemistry
| Species | Formula | Note |
| GLYX | CHOCHO | glyoxal |
| HCOOH | HCOOH | formic acid |
| IALD | HOCH2C(CH3)=CHCHO | hydroxy carbonyl alkenes from isoprene |
Reactions
New reactions
| No | Reaction | Rate Constant | Reference | Note |
|---|---|---|---|---|
| 1 | ATOOH +OH = ATO2 +H2O | 2.66E-12exp(200/T) | Rate uses MP+OH | |
| 2 | ATOOH +OH = MGLY +OH +H2O | 1.14E-12exp(200/T) | Rate uses MP+OH | |
| 3 | RIO2 + NO = 0.883NO2 + 0.783HO2 + 0.660CH2O + 0.400MVK + 0.260MACR + 0.070ISOPND + 0.047ISOPNB + 0.123HC5 + 0.1DIBOO | 2.7E-12 exp(350/T) | Paulot 2009a | HNO3 channel deleted since nitrate is treated explicitly;paulothn2009;neglect methylfuran formation (increase the yield of other products) |
| 4 | VRO2 + NO = 0.88NO2 + 0.35HO2 + 0.35CH2O + 0.53MCO3 + 0.53GLYC + 0.35MGLY + 0.12MVKN | 2.7E-12 exp(350/T) | Paulot 2009a | |
| 5 | MRO2 + NO = 0.85NO2 + 0.85HO2 + 0.122MGLY + 0.728HAC + 0.728CO + 0.122CH2O + 0.15MACRN | 2.7E-12 exp(350/T) | Paulot 2009a | This is modified based on Chuong et al. (2004).It was equally yield for MGLY and HAC in Paulot 2009 ACP, according to Peeters decomposition scheme. |
| 6 | MAN2 + NO = 1.5NO2 + 0.5CH2O + 0.5MGLY + 0.5PROPNN + 0.5CO + 0.5OH | 2.7E-12 exp(350/T) | Tyndall ETO2+NO | |
| 7 | ATO2 + HO2 = 0.15MCO3 + 0.15OH + 0.15CH2O + 0.85ATOOH | 8.60E-13 exp(700/T) | Dillon et al. (2008) | Tyndall, Dillon et al. (ACP 2008) cycling 15%,reduce the recyling by 5% compared to previous version to be fully consistent with Dillon et al. |
| 8 | KO2 + HO2 = 0.15OH +0.15ALD2 +0.15 MCO3 + 0.85 ATOOH | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=4 | MCM | assuming 15% recycling of OH |
| 9 | RIO2 + HO2= 0.88RIP + 0.12OH + 0.047MACR + 0.073MVK + 0.12HO2 + 0.12CH2O | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 | Paulot 2009b | Rate is from Saunders et al. (2003) |
| 10 | VRO2 + HO2 = 0.1VRP + 0.68OH + 0.578GLYC + 0.578MCO3 + 0.187MEK + 0.102HO2 + 0.102CH2O + 0.102MGLY + 0.033RCHO | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=4 | ??? | crounse2010 |
| 11 | MRO2 + HO2 = 1.0MRP | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=4 | isomerization of MRO2 is included in another reaction. | |
| 12 | MAN2 + HO2 = 0.075PROPNN + 0.075CO + 0.075HO2 + 0.075MGLY + 0.075CH2O + 0.075NO2 + 0.15OH + 0.85ISNP | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)],n=4 | assume 15% recycling of OH, the rest goes to ISNP | |
| 13 | INO2 + HO2 = INPN | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 | Xie et al. (2012) | See nighttime chemistry |
| 14 | RIO2 + MO2 = 1.1HO2 + 1.22CH2O + 0.280MVK + 0.180MACR + 0.3HC5 + 0.24MOH + 0.24ROH | 8.37E-14 | ||
| 15 | RIO2 + RIO2 = 1.28HO2 + 0.92CH2O + 0.56MVK + 0.36MACR + 0.48ROH + 0.5HC5 | 1.54E-13 | ||
| 16 | HC5OO + MO2 = 0.50HO2 + 0.33CO + 0.09H2 + 0.18HAC + 0.13GLYC + 0.29MGLY + 0.25MEK + 0.95CH2O + 0.25MOH + 0.25ROH + 0.5HO2 | 8.37E-14 | Tyndall MO2+MO2 Atkinson97 RO2+RO2; HC5OO=old IAO2 | |
| 17 | MRO2 + MO2 = 0.595HAC + 0.255MGLY + 0.595CO + 1.255CH2O + 1.7HO2 + 0.150ROH | 8.37E-14 | ||
| 18 | MAN2 + MO2 = 0.375PROPNN + 0.375CO + 0.375HO2 + 0.375MGLY + 0.375CH2O + 0.375NO2 + 0.250CH2O + 0.250R4N2 | 8.37E-14 | ||
| 19 | INO2 + MO2 = 0.55NO2 + 0.40HO2 + 0.425R4N2 + 0.025NO2 + 0.05MACR + 0.08CH2O + 0.03MVK + 0.25RCHO + 0.75CH2O + 0.25MOH + 0.25ROH + 0.5HO2 | 8.37E-14 | ||
| 20 | RCO3 + HO2 = 0.410RP + 0.150RCOOH + 0.150O3 + 0.440OH + 0.440ETO2 | 4.3E-13exp(1040/T) | MCM v3.2 | Branching ratio is from MCMv3.2 |
| 21 | MAO3 + HO2 = 0.44OH +0.15O3 + 0.59CH2O + 0.39MO2 + 0.41MAOP + 0.39CO | 4.3E-13exp(1040/T) | use MCM, 44% OH channel, 15% O3 channel, 41% peroxide channel. | |
| 22 | PMN + OH = HAC + CO + NO2 | 2.90E-11 | MCM v3.2 | rates and products all from MCM |
| 30 | GLYC + OH = 0.850CH2O +0.260CO2 + 0.580CO + 0.260OH + 0.740HO2 + 0.155GLYX | FRAC=1-11.0729*exp(-1/73T) Rate=8.00E-12*FRAC | Paulot 2009a | Butkovskaya 2006 companion paper and Paulot 2009 |
| 31 | GLYC + OH= HCOOH + 0.190OH +0.190CO + 0.810CO2 + 0.810HO2 | FRAC=1-11.0729*exp(-1/73T) Rate=8.00E-12*(1-FRAC) | Paulot 2009a | Butkovskaya 2006 companion paper and Paulot 2009 |
| 32 | GLYX+OH = HO2+2CO | 3.1E-12exp(340/T) | IUPAC2008 | JMAO |
| 33 | MACR + OH = 0.53MAO3 +0.47MRO2 | 8.0E-12exp(380/T) | Paulot 2009a | MAO3(=MCO3 in the paper); MRO2(=MACROO in the paper) |
| 34 | HAC + OH = MGLY +HO2 | FRAC=1-23.7*exp(1/60T) Rate=2.15E-12exp(305/T)*FRAC | Paulot 2009a | Butkovskaya JPC A (a,b)2006 and Paulot 2009a |
| 35 | HAC + OH = 0.5HCOOH + OH +0.5ACTA +0.5CO2 + 0.5CO + 0.5MO2 | FRAC=1-23.7*exp(1/60T) Rate=2.15E-12exp(305/T)*(1-FRAC) | Paulot 2009a | Butkovskaya JPC A (a,b)2006 and Paulot 2009a |
| 38 | PRPN+OH=0.209PRN1+0.791OH+0.791PROPNN | 8.78E-12exp(200/T) | Branching ratio is determined by alpha and beta Hydrogen position (Kwok et al., 1995). | |
| 39 | ETP+OH=0.64OH+0.36ETO2+0.60ALD2 | 5.18E-12exp(200/T) | Branching ratio is determined by alpha and beta Hydrogen position (Kwok et al., 1995). | |
| 40 | RA3P+OH=0.64OH+0.36A3O2+0.64RCHO | 5.18E-12exp(200/T) | Branching ratio is determined by alpha and beta Hydrogen position (Kwok et al., 1995). | |
| 41 | RB3P+OH=0.791OH+0.209B3O2+0.791ACET | 8.78E-12exp(200/T) | Branching ratio is determined by alpha and beta Hydrogen position (Kwok et al., 1995). | |
| 42 | R4P+OH=0.791OH+0.209R4O2+0.791RCHO | 8.78E-12exp(200/T) | Branching ratio is determined by alpha and beta Hydrogen position (Kwok et al., 1995). | |
| 43 | RP+OH=RCO3 | 6.13E-13exp(200/T) | same as MAP+OH | |
| 44 | PP+OH=0.791OH+0.209PO2+0.791HAC | 8.78E-12exp(200/T) | Branching ratio is determined by alpha and beta Hydrogen position (Kwok et al., 1995). | |
| 45 | RIP+OH = 0.387RIO2 + 0.613OH + 0.613HC5 | 4.75E-12exp(200/T) | Paulot 2009b | branching ratio is derived below |
| 46 | RIP + OH = OH + IEPOX | 1.9E-11exp(390/T) | Paulot 2009b | the yield of IEPOX is > 70% assumed to be 100% |
| 47 | IEPOX + OH = IEPOXOO | 5.78e-11exp(-400/T) | Paulot 2009b | |
| 48 | IEPOXOO + HO2 = 0.725HAC + 0.275GLYC + 0.275GLYX + 0.275MGLY + 1.125OH + 0.825HO2 + 0.200CO2 + 0.375CH2O + 0.074HCOOH + 0.251CO | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 | Paulot 2009b | |
| 49 | IEPOXOO +NO=0.725HAC+0.275GLYC+0.275GLYX +0.275MGLY +0.125OH +0.825HO2+0.200CO2+0.375CH2O +0.074HCOOH +0.251CO +NO2 | 2.7E-12exp(350/T) | FP: No peroxide was observed | |
| 50 | IAP+OH =0.654OH+0.654DHMOB+0.346HC5OO | 5.31E-12 exp(200/T) | ||
| 51 | VRP+OH=0.791OH+0.791MEK+0.209VRO2 | 8.78E-12exp(200/T) | ||
| 52 | MRP + OH = MRO2 | 1.84E-12exp(200/T) | This channel is for the abstraction of peroxide H (OOH), which is slow and ignored in MCM v3.2 | |
| 53 | MRP + OH = CO2 + HAC + OH | 4.40E-12exp(380/T) | This second channel is for the abstraction of aldehydic H, much faster! The rate is from MACR + OH. | |
| 54 | MAOP + OH = MAO3 | 6.13E-13exp(200/T) | same as MAP+OH | |
| 55 | MAOP + OH = MAOPO2 | 3.60E-12exp(380/T) | ||
| 56 | MCO3 + MAOPO2 = 1.0HAC + 2.0CO2 + OH + MO2 | 1.68E-12exp(500/T) | ||
| 57 | MCO3 + MAOPO2 = 1.0ACTA+1.0MEK | 1.87E-13 exp(500/T) | ||
| 58 | MAOPO2 + MO2 = 0.7HAC +0.7CO2+0.7OH+1.0CH2O+0.7HO2+0.3ROH | 8.37E-14 | ||
| 59 | MAOPO2 + MAOPO2 = 2.0HAC+2.0CO2+2.0OH | 8.37E-14 | ||
| 60 | MAOPO2 + HO2 = 1.0HAC+1.0CO2+2.0OH | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=4 | ||
| 61 | MAOPO2 + NO = 1.0HAC+1.0CO2+1.0OH+1.0NO2 | K* (1-YN) where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=4.00E00) | ||
| 62 | MAOPO2 + NO = 1.0HNO3 | K* YN where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=4.00E00) | Not treated explicitly | |
| 63 | OH + MAP = 1.0MCO3 | 6.13E-13exp(200/T) | From J. Orlando (unpublished results), how confident is this temperature dependence? | |
| 64 | HC5 + O3 = 0.6MGLY + 0.1OH + 0.12CH2O + 0.28GLYC + 0.3O3 + 0.4CO + 0.2H2 + 0.2HAC + 0.2HCOOH | 6.16E-15 exp(-1814/T) | HC5=old IALD?? | |
| 65 | R4O2 + MCO3 = MO2 + 0.32ACET + 0.19MEK + 0.27HO2 + 0.32ALD2 + 0.13RCHO + 0.05A3O2 + 0.18B3O2 + 0.32ETO2 | 1.68E-12 exp(500/T) | ||
| 66 | ATO2+MCO3 = MCO3 +CH2O +MO2 | 1.68E-12 exp(500/T) | IUPAC06 | |
| 67 | RIO2 + MCO3 = 0.887HO2 + 0.747CH2O + 0.453MVK + 0.294MACR + 0.140HC5 + 0.113DIBOO + CO2 + MO2 | 1.68E-12 exp(500/T) | Follow RIO2+NO without the yield of nitrate and NO2 and then rescale it. | |
| 68 | HC5OO + MCO3 = 0.216GLYX + 0.234MGLY + 0.234GLYC + 0.216HAC + 0.29DHMOB + 0.17MOBA + 0.09RCHO + HO2 + 0.09CO + MO2 | 1.68E-12 exp(500/T) | HC5OO=old IAO2, this radical channel use the same as HC5OO+NO without NO2 yield. | |
| 69 | VRO2 + MCO3 = 0.4HO2 + 0.4CH2O + 0.6MCO3 + 0.6GLYC + 0.4MGLY + 1.0MO2 | 1.68E-12 exp(500/T) | this radical channel use the same as VRO2+NO without NO2 and MVKN yield. And carbon balance. | |
| 70 | MRO2 + MCO3 = 0.850HO2 + 0.143MGLY + 0.857HAC + 0.857CO + 0.143CH2O +1.0MO2 | 1.68E-12 exp(500/T) | this radical channel use the same as MRO2+NO without NO2 and MACRN yield. | |
| 71 | MAN2+MCO3 = MO2 +.5NO2+.5CH2O+.5MGLY+.5PROPNN+.5CO+.5HO2 | 1.68E-12 exp(500/T) | ||
| 72 | ATO2 +MCO3 =MGLY+ACTA | 1.87E-13exp(500/T) | IUPAC06 | replace MEK with MGLY |
| 73 | HC5OO+MCO3 = MEK +ACTA | 1.87E-13 exp(500/T) | ||
| 74 | ISOPNB + O3 = 0.610MVKN + 0.390MACRN + 0.27OH + CH2O | 1.06E-16 | Lockwood et al., 2010 ACP | use 1,2 for beta channel |
| 75 | ISOPND + O3 = 0.5PROPNN + 0.5ETHLN + 0.27OH + 0.5GLYC + 0.5HAC | 5.3E-17 | Lockwood et al., 2010 ACP | use 1,4 for delta channel |
| 76 | HC5+OH=HC5OO | 3.35E-11exp(380/T) | Paulot 2009a | |
| 77 | HC5OO + NO = NO2 + 0.216GLYX + 0.234MGLY + 0.234GLYC + 0.216HAC + 0.290DHMOB + 0.170MOBA + 0.090RCHO + HO2 + 0.090CO | K* (1-YN) where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=4.00E00) | ||
| 78 | HC5OO +NO=HNO3 | K* YN where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=5.00E00) | ||
| 79 | HC5OO + HO2 = 0.1IAP + 0.9OH + 0.9MGLY + 0.9GLYC + 0.9HO2 | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 | 90% recycling, no experimental data. Somewhat based upon the high recycling rate observed for MVK/MACR | |
| 80 | ISOPND+OH=ISOPNDO2 | 2.64E-11exp(380/T) | Paulot 2009a | |
| 81 | ISOPNB+OH=ISOPNBO2 | 3.61E-12exp(380/T) | Paulot 2009a | |
| 82 | ISOPNDO2 + NO = 0.070MACRN + 0.310HCOOH + 0.440HAC + 0.130ETHLN + 0.650CH2O + 1.340NO2 + 0.150GLYC + 0.310NO3 + 0.150PROPNN + 0.340MEK + 0.350HO2 | K* (1-YN) where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=4.00E00) | Paulot 2009a | |
| 83 | ISOPNDO2+NO=HNO3 | K* YN where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=5.00E00) | Nitrates from ISOPND could not be observed in this experiment | |
| 84 | ISOPNDO2 + HO2 = 0.035MACRN + 0.155HCOOH + 0.22HAC + 0.065ETHLN + 0.325CH2O + 0.170NO2 + 0.075GLYC + 0.155NO3 + 0.075PROPNN + 0.170MEK + 0.175HO2 + 0.5OH + 0.5ISNP | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 | Assume 50% recycling from HO2+RO2 (no experimental data - check Ng et al. for better estimates) | |
| 85 | ISOPNBO2 + NO = 0.6GLYC + 0.6HAC + 0.4CH2O + 1.6NO2 + 0.26MACRN + 0.4HO2 + 0.14MVKN | K* (1-YN) where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=4.00E00) | Paulot 2009a | |
| 86 | ISOPNBO2 + NO = HNO3 | K* YN where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=5.00E00) | Nitrates from ISOPND could not be observed in this experiment | |
| 87 | ISOPNBO2 + HO2 = 0.3GLYC + 0.3HAC + 0.2CH2O + 0.13MACRN + 0.07MVKN + 0.3NO2 + 0.2HO2 + 0.5OH + 0.5ISNP | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 | Assume 50% recycling from HO2+RO2 (no experimental data - check Ng et al. for better estimates) | |
| 88 | ISNP+OH=0.612OH+0.612R4N1++0.193ISOPNBO2+0.193ISOPNDO2 | 4.75E-12exp(200/T) | replace the old ISNP+OH | |
| 89 | MVKN+OH=0.650HCOOH+NO3+0.650MGLY+0.350CH2O+0.350PYAC | 1.5E-12exp(380/T) | Paulot 2009a | |
| 90 | MACRN + OH = 1.0MACRNO2 | 1.39E-11exp(380/T) | ||
| 91 | MACRNO2 + NO = 0.08ACTA + 0.08CH2O + 0.15NO3 + 0.07HCOOH + 0.070MGLY + 0.850HAC + 0.85NO2 + 0.93CO2 + 1.0NO2 | 2.7E-12exp(350/T) | no nitrate yield (acyl) | |
| 92 | MACRNO2 + HO2 = 0.08ACTA + 0.08CH2O + 0.15NO3 + 0.07HCOOH + 0.07MGLY + 0.85HAC + 0.85NO2 + 0.93CO2 + 1.0OH | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=4 | Assume 100% recycling. No experiment data. Inferred from the very high recycling observed for MAO2+HO2 | |
| 93 | MACRNO2 + NO2=1.0PMNN | LPL: 9.00E-28(300/T)^8.9 HPL:7.70E-12(300/T)^0.2 Fc: 0.6 | ||
| 94 | PMNN =1.000MACRNO2 +1.000NO2 | 9e-29*exp(14000/T) | ||
| 95 | DHMOB + OH = 1.5CO + 1.0HO2 + 0.5HAC + 0.5MEK | 2.52E-11exp(410/T) | ||
| 96 | DIBOO +NO=HO2+NO2+0.520GLYC +0.520MGLY +0.480HAC+0.480GLYX | K* (1-YN) where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=4.00E00) | Dibble, 2004 Note that the yield of DIBOO Is likely overestimate (~30%) | |
| 97 | DIBOO +NO=HNO3 | K* YN where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=5.00E00) | ||
| 98 | DIBOO + HO2 = 0.15HO2 + 0.15OH + 0.078GLYC + 0.078MGLY + 0.072HAC + 0.072GLYX + 0.85R4P | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 | Assume 100% recycling. No experimental data. Somewhat derived from the high recycling in MVK/MACR | |
| 99 | MOBA +OH=MOBAOO | 2.79E-11exp(380/T) | ||
| 100 | MOBAOO+NO=RCHO+CO2+HO2+NO2 | K* (1-YN) where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=4.00E00) | ||
| 101 | MOBAOO+NO=HNO3 | K* YN where YN is returned from fyrno3.f K=2.7E-12 exp(350/T) (Xcarbn=5.00E00) | ||
| 102 | MOBAOO+HO2=0.500OH+0.500HO2+0.500RCHO+0.500CO2+0.500R4P | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 | Paulot 2009a | No experimental constraint. Assume 50% recycling and go to R4P for the peroxide channel to avoid carrying another peroxide |
| 103 | MOBA +O3=OH +HO2+CO2+MEK | 2.00E-17 | Paulot 2009a | Weak constraint on the rate constant - no constraint on the products |
| 104 | ETHLN +OH=CH2O +CO2+NO2 | 1.00E-11 | ||
| 105 | PROPNN+OH=NO2+MGLY | 1.00E-15 | Paulot 2009a | IUPAC says < 1e-12;Experiment suggests it is slower than than 1e-13-1e-15 |
| 106 | RIO2 = 2.0HO2 + 1.0CH2O + 0.5MGLY + 0.5GLYC + 0.5GLYX + 0.5GLYX + 0.500HAC + 1.0OH | 4.07E+08 exp(-7694/T) | Peeters et al. (2009, 2010) | Isomerization rate is adjusted according to Crounse et al. (2010), products follow Stavrakou et al. (2010). |
| 107 | MRO2 = 1.0CO + 1.0HAC + 1.0OH | 2.90E+07 exp(-5297/T) | Crounse et al. (2012) | 1,4-H-shift isomerization rate dominates over 1,5-H-shift. |
| 108 | ISOP + O3 = 0.244MVK + 0.325MACR + 0.845CH2O + 0.110H2O2 + 0.522CO + 0.204HCOOH + 0.199MCO3 + 0.026HO2 + 0.270OH + 0.128PRPE + 0.051MO2 | 1.00E-14 *EXP(-1970/T) | MCM v3.2 | rate is from JPL 11, products from MCM, assuming CH2OO is dominated by reactions with H2O. ISOP + O3 in standard chem is not carbon-balanced. |
| 109 | MVK + O3 = 0.202OH + 0.202HO2 + 0.352HCOOH + 0.535CO + 0.050ALD2 + 0.950MGLY + 0.050CH2O | 8.5 E-16exp(-1520/T) | MCM? | Rate is from IUPAC06 |
| 110 | MACR + O3 = 0.261OH + 0.202HO2 + 0.326HCOOH + 0.569CO + 0.880MGLY + 0.120CH2O | 1.4 E-15exp(-2100/T) | MCM? | |
| Photolysis | RIP = OH + HO2 + 0.710CH2O + 0.425MVK + 0.285MACR + 0.29HC5 | |||
| Photolysis | MACRN=NO2+HAC+MGLY+0.500CH2O+HO2+0.500CO | |||
| Photolysis | MVKN=GLYC +NO2+MCO3 | |||
| Photolysis | ISOPNB=HC5+NO2+HO2 | |||
| Photolysis | ISOPND=HC5+NO2+HO2 | |||
| Photolysis | PROPNN=CH2O +NO2+CO +MO2 | |||
| Photolysis | ATOOH=OH +CH2O +MCO3 |
RIP+OH
Follow SAR rules assuming a C(OOH) = 2* C(OH) = 7 (for the abstraction of the H alpha of the peroxide group). (see Kwok 1995 paper)
Assume that the abstraction of the peroxide H has a constant rate @298K of 3.6e-12
This gives for RIP:
43% 3.6e-12 (1,2) 28% 3.6e-12+7*1.94e-12 (4,3) 29% 3.6e-12+7*0.937e-12 (1,4)+(4,1) (I neglected 3,4 and 2,1) 9.3e-12@298K (4.75e-12*exp(200/T)) 0.387 not recycling
Therefore 0.387=3.6/9.3. RIP + OH = 0.387RIO2 + 0.613OH + 0.613HC5.
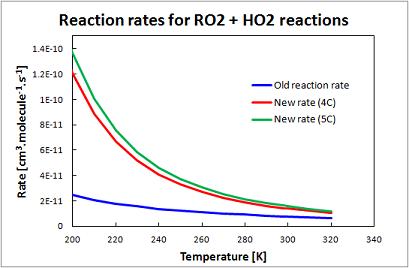
Update One - RO2+HO2 Reaction Rate
Update applied to all >C2 RO2 species reaction with HO2. These include, in the standard scheme, R4O2, R4N1, KO2, RIO2, RIO1, IAO2,ISN1, VRO2, MRO2, MVN2, MAN2, B3O2, INO2, PRN1, A3O2, PO2.
Old RO2+HO2 reaction rate: k = 7.40E-13*EXP(700/T)
New RO2+HO2 reaction rate: k = 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], where n=no. of carbon atoms
Comparison of new and old RO2+HO2 reaction rates for C4 RO2 and C5 RO2:
Benchmarking for this update can be viewed at: (Benchmarking results for RO2+HO2 reaction)
Update Two - Transport of RIP
RIP = isoprene peroxide species formed at low-NOx (i.e. via the RO2+HO2 pathway)
This benchmark is done to understand the muted influence of the increased rate of the RO2+HO2 reaction on CH2O. Is this because the ultimate yield of CH2O is similar for all levels of NOx and RIP is not transported, leading to the realization of the ultimate yield of CH2O in the same grid box as its emission source?
Both schemes are run with initial concentrations of species set to zero. As RIP is added as an additional transported species this was viewed as the most effective way of comparing the two model runs.
Benchmarking for this update can be viewed at: (Benchmarking results for transporting RIP)
Updates 02/04/2013
| Old reaction | Old rate | New reaction | New rate | Note |
|---|---|---|---|---|
| KO2+HO2 =OH +ALD2 +MCO3 | 7.40E-13 exp(700/T) | KO2 + HO2 = 0.15OH +0.15ALD2 +0.15 MCO3 + 0.85 ATOOH | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=4 | Assuming 15% recycling of OH, consistent with MCM. Rate is also from Saunders et al. (2003). |
| MRO2 +HO2 =0.020MRP+0.980OH +0.980HO2+0.294CH2O+0.686HAC +0.294MGLY +0.686CO | 7.40E-13 exp(700/T) | MRO2 + HO2 = 1.0MRP | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=4 | Isomerization of MRO2 is already taken into account in another reaction. |
| MAN2 + HO2 = 0.5PROPNN + 0.5CO + 0.5HO2 + 0.5MGLY + 0.5CH2O + 0.5NO2 + OH | 7.40E-13 exp(700/T) | MAN2 + HO2 = 0.075PROPNN + 0.075CO + 0.075HO2 + 0.075MGLY + 0.075CH2O + 0.075NO2 + 0.15OH + 0.85ISNP | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=4 | assuming 15% recycling |
| INO2 + HO2 = 0.5INPN + 0.5ISOPND + 0.5OH + 0.5HO2 | 7.40E-13 exp(700/T) | INO2 + HO2 = INPN | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 | See nighttime chemistry |
| MAO3 + HO2 = 0.87OH +0.03O3 + 0.435CH2O + 0.435MO2 + 0.1MAOP + 0.030RCOOH + 0.283HAC + 0.152ATO2 + 0.870CO2 + 0.435CO | 4.3E-13exp(1040/T) | MAO3 + HO2 = 0.44OH +0.15O3 + 0.59CH2O + 0.39MO2 + 0.41MAOP + 0.39CO | 4.3E-13exp(1040/T) | use MCM, 44% OH channel, 15% O3 channel, 41% peroxide channel. |
| PMN + OH = 1.000PMNO2 | 3.20E-11 | PMN + OH = HAC + CO + NO2 | 2.90E-11 | from MCM |
| PMNO2 + NO = 0.6CO2 + 0.6HAC + 0.6NO3 + 0.4CH2O + 0.4HO2 + 0.4PYPAN + 0.900NO2 | K* (1-YN) where YN isreturned from fyrno3.f; K=2.7E-12 exp(350/T) (Xcarbn=4.0E00) | we now remove all reactions from PMN following MCM. | ||
| PMNO2 + NO=PMNN | K* YN where YN is returned from fyrno3.f ; K=2.7E-12 exp(350/T) (Xcarbn=4.0E00) | |||
| PMNO2 + HO2 = 0.6CO2 + 0.6HAC + 0.6NO3 + 0.4CH2O + 0.4HO2 + 0.4PYPAN + 0.5R4P + 0.5OH | 7.4E-13exp(700/T) | |||
| PYPO2 + NO2 + M = PYPAN | LPL: 9.0E-28(300/T)^8.9 HPL:7.70E-12(300/T)^0.2 Fc: 0.6 | |||
| PYPAN =PYPO2 +NO2 | 9.0E-29exp(14000/T) | |||
| PYPO2 + NO = CO2+MCO3 +NO2 | 2.7E-12 exp(350/T) | |||
| PYPO2 + HO2 = CO2+MCO3 +OH | 7.40E-13 exp(700/T) | |||
| PYPAN = 0.300NO3+0.700NO2+MCO3 +CO2 | photolysis | |||
| PYPAN = NO3 + MCO3 + CO2 | photolysis | |||
| PP+OH=0.791OH+0.209PO2+0.791RCHO | 8.78E-12exp(200/T) | PP+OH=0.791OH+0.209PO2+0.791HAC | ||
| INPN+OH= 1.0OH + 1.0NO2 + 1.0MEK | 1.9E-11exp(390/T) | Now replaced by new nighttime chemistry | ||
| INPN+OH=0.36INO2+0.64R4N2+0.64OH | 5.18E-12exp(200/T) | Now replaced by new nighttime chemistry | ||
| DIBOO+HO2 = HO2 + OH + 0.52GLYC + 0.52MGLY + 0.48HAC + 0.48GLYX | 7.4E-13exp(700/T) | DIBOO + HO2 = 0.15HO2 + 0.15OH + 0.078GLYC + 0.078MGLY + 0.072HAC + 0.072GLYX + 0.85R4P | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 | assume 15% recycling of OH, rest goes to R4P |
| MOBAOO + HO2 = 0.5OH + 0.5HO2 + 0.5RCHO + 0.5CO2 + 0.5R4P | 7.4E-13exp(700/T) | MOBAOO + HO2=0.15OH + 0.15HO2 + 0.15RCHO + 0.15CO2 + 0.85R4P | 2.91E-13*EXP(1300/T)[1-EXP(-0.245*n)], n=5 | assume 15% recycling of OH, rest goes to R4P |
References
- Butkovskaya, N. I., Pouvesle, N., Kukui, A., and Le Bra, G.: Mechanism of the OH-initiated oxidation of glycolaldehyde over the temperature range 233-296 K, J. Phys. Chem. A, 110, 13492-13499, 10.1021/jp064993k, 2006a.
- Butkovskaya, N. I., Pouvesle, N., Kukui, A., Mu, Y. J., and Le Bras, G.: Mechanism of the OH-initiated oxidation of hydroxyacetone over the temperature range 236-298 K, J. Phys. Chem. A, 110, 6833-6843, 10.1021/jp056345r, 2006b.
- Chuong, B., and Stevens, P.: Measurements of the kinetics of the OH-initiated oxidation of methyl vinyl ketone and methacrolein, Int. J. Chem. Kinet., 36, 12-25, 2004.
- Crounse, J. D., Paulot, F., Kjaergaard, H. G., and Wennberg, P. O.: Peroxy radical isomerization in the oxidation of isoprene, Phys. Chem. Chem. Phys., 13, 13607-13613, 10.1039/C1CP21330J 2011.
- Crounse, J. D., Knap, H. C., Ørnsø, K. B., Jørgensen, S., Paulot, F., Kjaergaard, H. G., and Wennberg, P. O.: Atmospheric Fate of Methacrolein. 1. Peroxy Radical Isomerization Following Addition of OH and O2, The Journal of Physical Chemistry A, 116, 5756-5762, 10.1021/jp211560u, 2012.
- Dillon, T. J., and Crowley, J. N.: Direct detection of OH formation in the reactions of HO2 with CH3C(O)O-2 and other substituted peroxy radicals, Atmos. Chem. Phys., 8, 4877-4889, 2008.
- Ito, A., Sillman, S., and Penner, J. E.: Effects of additional nonmethane volatile organic compounds, organic nitrates, and direct emissions of oxygenated organic species on global tropospheric chemistry, J. Geophys. Res.-Atmos., 112, 10.1029/2005jd006556, 2007.
- Jenkin, M. E., Saunders, S. M., and Pilling, M. J.: The tropospheric degradation of volatile organic compounds: a protocol for mechanism development, Atmos. Environ., 31, 81-104, http://dx.doi.org/10.1016/S1352-2310(96)00105-7, 1997.
- Kwok, E. S. C., and Atkinson, R.: Estimation of hydroxyl radical reaction-rate constants for gas-phase organic-compounds using a structure-reactivity relationship - an update, Atmos. Environ., 29, 1685-1695, 10.1016/1352-2310(95)00069-b, 1995.
- Lockwood, A. L., Shepson, P. B., Fiddler, M. N., and Alaghmand, M.: Isoprene nitrates: preparation, separation, identification, yields, and atmospheric chemistry, Atmos. Chem. Phys., 10, 6169-6178, 10.5194/acp-10-6169-2010, 2010.
- Marais, E. A., Jacob, D. J., Kurosu, T. P., Chance, K., Murphy, J. G., Reeves, C., Mills, G., Casadio, S., Millet, D. B., Barkley, M. P., Paulot, F., and Mao, J.: Isoprene emissions in Africa inferred from OMI observations of formaldehyde columns, Atmos. Chem. Phys., 12, 6219-6235, 10.5194/acp-12-6219-2012, 2012.
- Master Chemical Mechanism, MCM v3.2 (Jenkin et al., Atmos. Environ., 31, 81, 1997; Saunders et al., Atmos. Chem. Phys., 3, 161, 2003), via website: http://mcm.leeds.ac.uk/MCM.
- Paulot, F., Crounse, J. D., Kjaergaard, H. G., Kroll, J. H., Seinfeld, J. H., and Wennberg, P. O.: Isoprene photooxidation: new insights into the production of acids and organic nitrates, Atmos. Chem. Phys., 9, 1479-1501, 2009a.
- Paulot, F., Crounse, J. D., Kjaergaard, H. G., Kurten, A., St Clair, J. M., Seinfeld, J. H., and Wennberg, P. O.: Unexpected Epoxide Formation in the Gas-Phase Photooxidation of Isoprene, Science, 325, 730-733, 10.1126/science.1172910, 2009b.
- Peeters, J., Nguyen, T. L., and Vereecken, L.: HOx radical regeneration in the oxidation of isoprene, Phys. Chem. Chem. Phys., 11, 5935-5939, 10.1039/b908511d, 2009.
- Peeters, J., and Müller, J. F.: HOx radical regeneration in isoprene oxidation via peroxy radical isomerisations. II: experimental evidence and global impact, Phys. Chem. Chem. Phys., 12, 14227-14235, 10.1039/c0cp00811g, 2010.
- Saunders, S. M., Jenkin, M. E., Derwent, R. G., and Pilling, M. J.: Protocol for the development of the Master Chemical Mechanism, MCM v3 (Part A): tropospheric degradation of non-aromatic volatile organic compounds, Atmos. Chem. Phys., 3, 161-180, 2003.
- Sander, S. P., J. Abbatt, J. R. Barker, J. B. Burkholder, R. R. Friedl, D. M. Golden, R. E. Huie, C. E. Kolb, M. J. Kurylo, G. K. Moortgat, V. L. Orkin and P. H. Wine "Chemical Kinetics and Photochemical Data for Use in Atmospheric Studies, Evaluation No. 17," JPL Publication 10-6, Jet Propulsion Laboratory, Pasadena, 2011 http://jpldataeval.jpl.nasa.gov.
- Stavrakou, T., Peeters, J., and Muller, J. F.: Improved global modelling of HOx recycling in isoprene oxidation: evaluation against the GABRIEL and INTEX-A aircraft campaign measurements, Atmos. Chem. Phys., 10, 9863-9878, 10.5194/acp-10-9863-2010, 2010.
- Xie, Y., Paulot, F., Carter, W. P. L., Nolte, C. G., Luecken, D. J., Hutzell, W. T., Wennberg, P. O., Cohen, R. C., and Pinder, R. W.: Understanding the impact of recent advances in isoprene photooxidation on simulations of regional air quality, Atmos. Chem. Phys. Discuss., 12, 27173-27218, 10.5194/acpd-12-27173-2012, 2012.